Publications
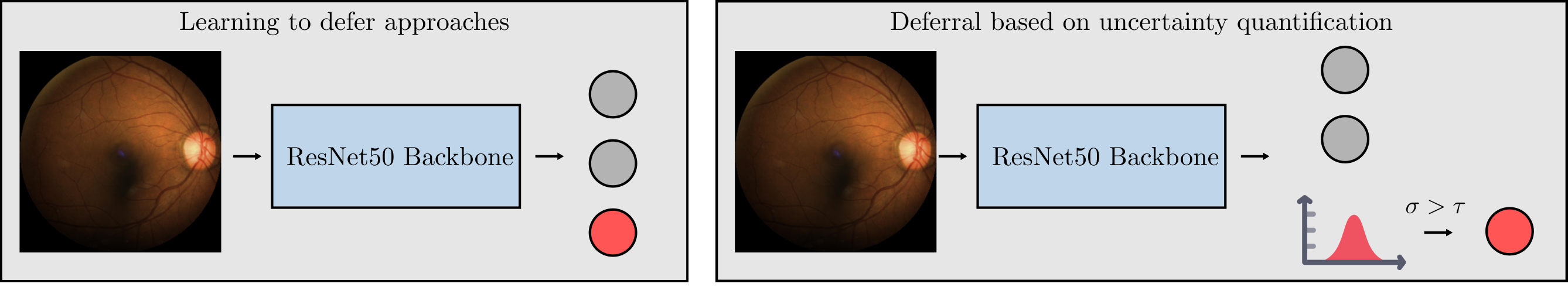
Is Uncertainty Quantification a Viable Alternative to Learned Deferral?
International Workshop on Uncertainty for Safe Utilization of Machine Learning in Medical Imaging (UNSURE) held in conjunction with MICCAI — 2025
Artificial Intelligence (AI) holds the potential to dramatically improve patient care. However, it is not infallible, necessitating human-AI-collaboration to ensure safe implementation. One aspect of AI safety is the models' ability to defer decisions to a human expert when they are likely to misclassify autonomously. Recent research has focused on methods that learn to defer by optimising a surrogate loss function that finds the optimal trade-off between predicting a class label or deferring. However, during clinical translation, models often face challenges such as data shift. Uncertainty quantification methods aim to estimate a model's confidence in its predictions. However, they may also be used as a deferral strategy which does not rely on learning from specific training distribution. We hypothesise that models developed to quantify uncertainty are more robust to out-of-distribution (OOD) input than learned deferral models that have been trained in a supervised fashion. To investigate this hypothesis, we constructed an extensive evaluation study on a large ophthalmology dataset, examining both learned deferral models and established uncertainty quantification methods, assessing their performance in- and out-of-distribution. Specifically, we evaluate their ability to accurately classify glaucoma from fundus images while deferring cases with a high likelihood of error. We find that uncertainty quantification methods may be a promising choice for AI deferral.
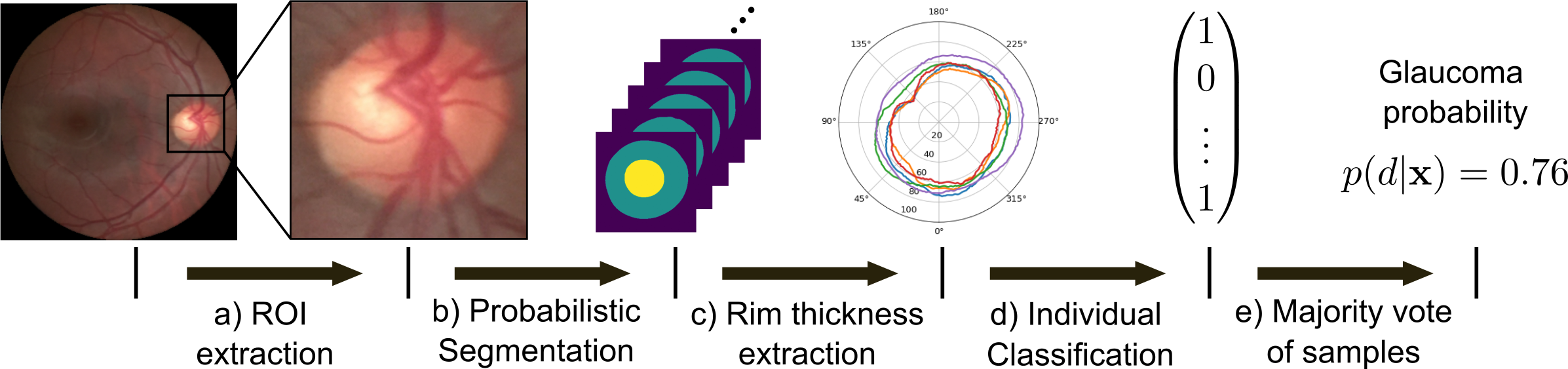
Leveraging Probabilistic Segmentation Models for Improved Glaucoma Diagnosis: A Clinical Pipeline Approach
Medical Imaging with Deep Learning (MIDL) — 2024
The accurate segmentation of the optic cup and disc in fundus images is essential for diagnostic processes such as glaucoma detection. The inherent ambiguity in locating these structures often poses a significant challenge, leading to potential misdiagnosis. To model such ambiguities, numerous probabilistic segmentation models have been proposed. In this paper, we investigate the integration of these probabilistic segmentation models into a multistage pipeline closely resembling clinical practice. Our findings indicate that leveraging the uncertainties provided by these models substantially enhances the quality of glaucoma diagnosis compared to relying on a single segmentation only.
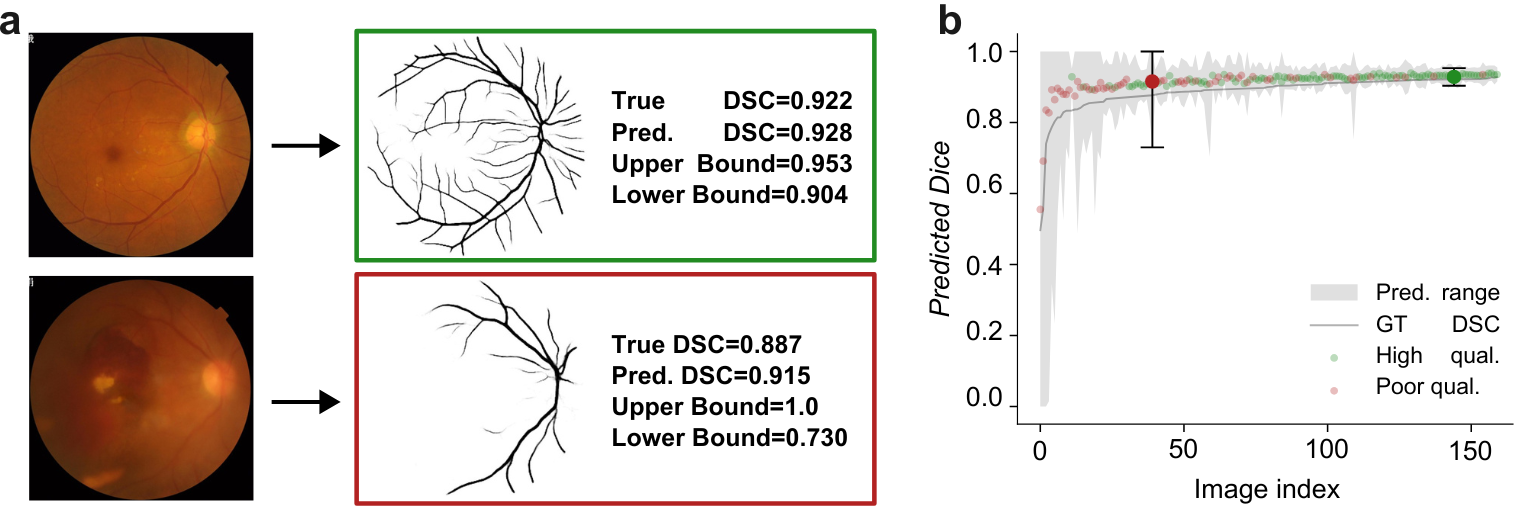
Conformal Performance Range Prediction for Segmentation Output Quality Control
International Workshop on Uncertainty for Safe Utilization of Machine Learning in Medical Imaging (UNSURE) held in conjunction with MICCAI — 2024
Recent works have introduced methods to estimate segmentation performance without ground truth, relying solely on neural network softmax outputs. These techniques hold potential for intuitive output quality control. However, such performance estimates rely on calibrated softmax outputs, which is often not the case in modern neural networks. Moreover, the estimates do not take into account inherent uncertainty in segmentation tasks. These limitations may render precise performance predictions unattainable, restricting the practical applicability of performance estimation methods. To address these challenges, we develop a novel approach for predicting performance ranges with statistical guarantees of containing the ground truth with a user specified probability. Our method leverages sampling-based segmentation uncertainty estimation to derive heuristic performance ranges, and applies split conformal prediction to transform these estimates into rigorous prediction ranges that meet the desired guarantees. We demonstrate our approach on the FIVES retinal vessel segmentation dataset and compare five commonly used sampling-based uncertainty estimation techniques. Our results show that it is possible to achieve the desired coverage with small prediction ranges, highlighting the potential of performance range prediction as a valuable tool for output quality control.